New product development (NPD) is spread with risks, at all stages of the product life cycle. Successful new product development is the art of managing design risks. The risks impact tightly balanced schedules, resources and costs that determine whether a product will launch on time while meeting performance and budget goals. The key to success is to identify and mitigate risks throughout development.
By following the best practices below, you can better manage your NPD risks.
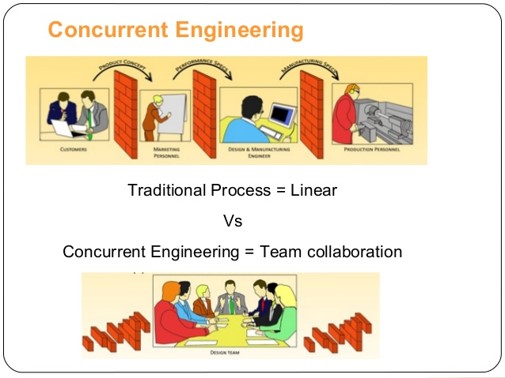
Best Practice #1: Concurrent Engineering
Concurrent engineering replaces the traditional “waterfall” development flow with an integrated “team” model, characterized by a cross-functional team driven by a project manager who owns the project. Design activities occur in parallel so that dependencies, risks and errors may be understood and mitigated as early as possible in the development cycle. A strong concurrent engineering team includes representatives from Marketing, Engineering, Operations, Finance, Service & Support.
Best Practice #2: Mitigate Risks
There are risks throughout the product development cycle, both known and emerging. Successful new product developers mitigate risk on an ongoing basis by
- focusing on basic product requirements and features;
- performing feasibility studies early;
- developing a product roadmap and setting realistic timelines;
- using risk assessment tools such as FMEA (“Failure Mode and Effect Analysis”);
- incorporating a strong NPI (“New Product Introduction”) process.
Best Practice #3: Employ Design For Excellence (DFX)
DFX methodologies underlie high quality products that are cost effectively built, tested, shipped and supported. Leverage DFX methodologies during key development transition points:
- Concept → (DFX) → Engineering Verification Test
- Engineering Verification Test → (DFX) → Design Verification Test
- Design Verification Test → (DFX) → Process Verification Test
- Process Verification Test → (DFX) → Volume Manufacturing
Best Practice #4: Leverage Rapid Prototyping

Use rapid prototyping to iterate quickly upon your design. Begin with a design based on your minimum viable product (“MVP”). Use market feedback to iterate on your design. Rapid prototyping techniques like simple mock-ups, 3D computer-aided design models, use of standard developer kits, and 3D printing allow you to test assumptions earlier in the development cycle. Here are just a few ways you can leverage this amazing method:
- Shape analysis for fit, ergonomics, appearance
- “Disposable” assembly tooling (when they wear out, just grow more)
- Component fitting for both internal and external enclosure designs
- Air flow testing for cooling or other functional aspects
- Mechanism concept testing (normally a very time consuming process)
Best Practice #5: Adhere to Agency & Environmental Compliance
Understand the regulatory environment and industry standards that governs your product prior to development, and develop a strategy to gain compliance. For example, for industries like medical devices, it is critical that you document product requirements from the outset to gain FDA compliance. Some typical agency & environmental compliance requirements include:
- Federal Drug Administration (FDA)
- Underwriters Laboratory (UL)
- European Conformity (CE)
- Federal Communications Commission (FCC)
- Evaluation, Authorization and Restriction of Chemical substances (REACH)
- Restriction of Hazardous Substances (RoHS)
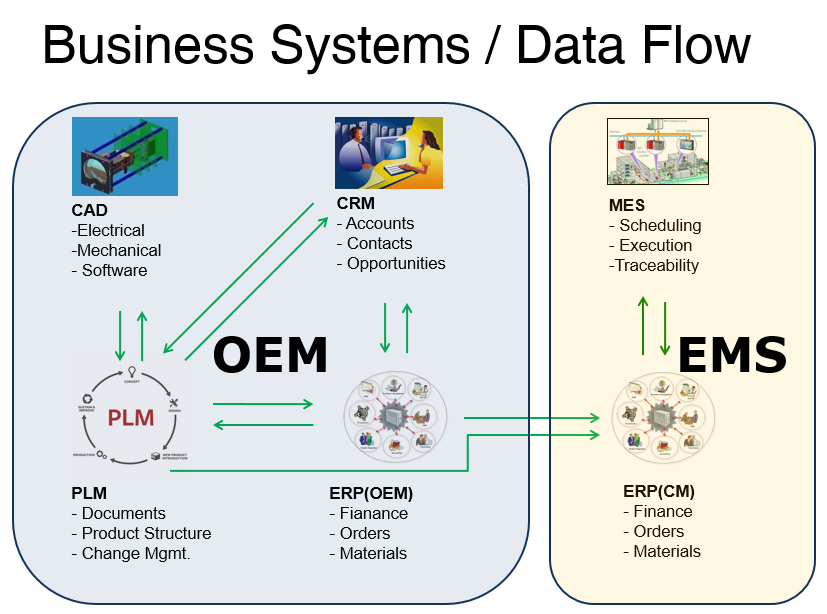
Best Practice #6: Deploy Scalable Business Systems
A cross-functional team enables strong communications — you also need strong business process and systems to support the team, scale the product, and maintain clarity between the product team, customers, OEMs, and other partners such as ODMs and Suppliers. Systems to deploy include Computer Aided Design (CAD), Customer Relationship Management (CRM), Product Lifecycle Management (PLM), Enterprise Resource Planning (ERP), Manufacturing Execution Systems (MES) and Quality Management Systems (QMS). See below for business systems and data flows:
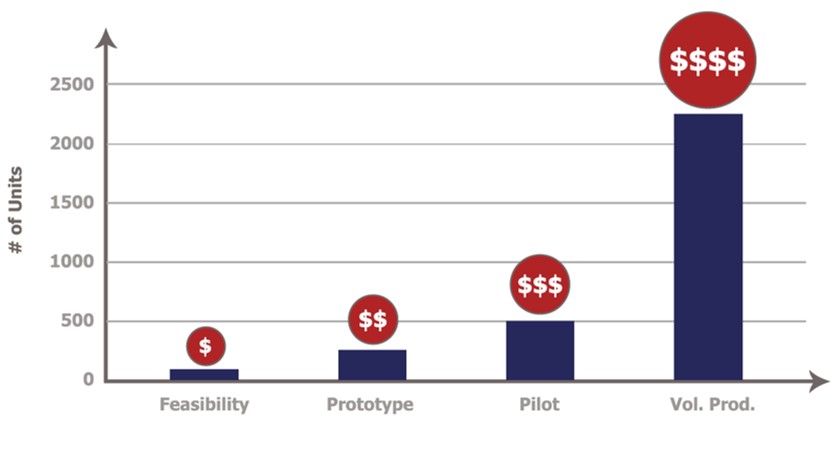
Best Practice #7: Learn From Prototypes & Early Builds
The costs to undo mistakes escalate the later the mistake is discovered. Using best practices, implementing reviews and mitigating risks that are identified in planned feasibility reviews, prototype builds, and pilots can reduce or eliminate design risks in the transition to volume production.
Cost Escalation through the Product Lifecycle